 
Chemical Engineering Research Information Center. (eds.) NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD) ( sample table of physical properties) ^ a b c "Binary Vapor-Liquid Equilibrium Data" (Queriable database). A substance in solid phase has low entropy in liquid phase, it has medium entropy in gas phase, it has high entropy.Archived from the original (Queriable database) on. Advanced Industrial Science and Technology. Solve the equation (specific heat at constant pressure Cp 4.1818 kJ/Kkg). Entropy of vaporization Data from NIST Standard Reference Database 69: NIST Chemistry WebBook The National Institute of Standards and Technology (NIST) uses its best efforts to deliver a high quality copy of the Database and to verify that the data contained therein have been selected on the basis of sound scientific judgment. Define final and initial temperature: Tf 20 ☌, Ti 100 ☌. of the heat of vaporization and the entropy of the liquid, as well as for the proper treatment of data on the vapor pressures of solutions of He and He4. ^ a b c "Spectral Database for Organic Compounds". We will use the change in entropy formula: s Cp × ln (Tf / Ti), where Tf and Ti indicate the final and the initial temperature, respectively.^ Lange's Handbook of Chemistry 10th ed, pp. 1522–1524.^ a b CRC Handbook of Chemistry and Physics 44th ed.^ a b Lange's Handbook of Chemistry 10th ed, pp. 1525–1528. It states that the entropy of vaporization at one atmospheric pressure of many liquids is almost constant: Table 1 shows a few values of the entropy of."Molar Heat Capacity (cP) of Methanol from Dortmund Data Bank". Note: Enthalpy of vaporization is the amount of energy that must be added to the liquid substance, to transform a quantity of that substance into gas. ^ "Vapor Pressure of Methanol from Dortmund Data Bank". We expect the entropy of a solid to be less than that of its liquid to be less than that of its vapour. 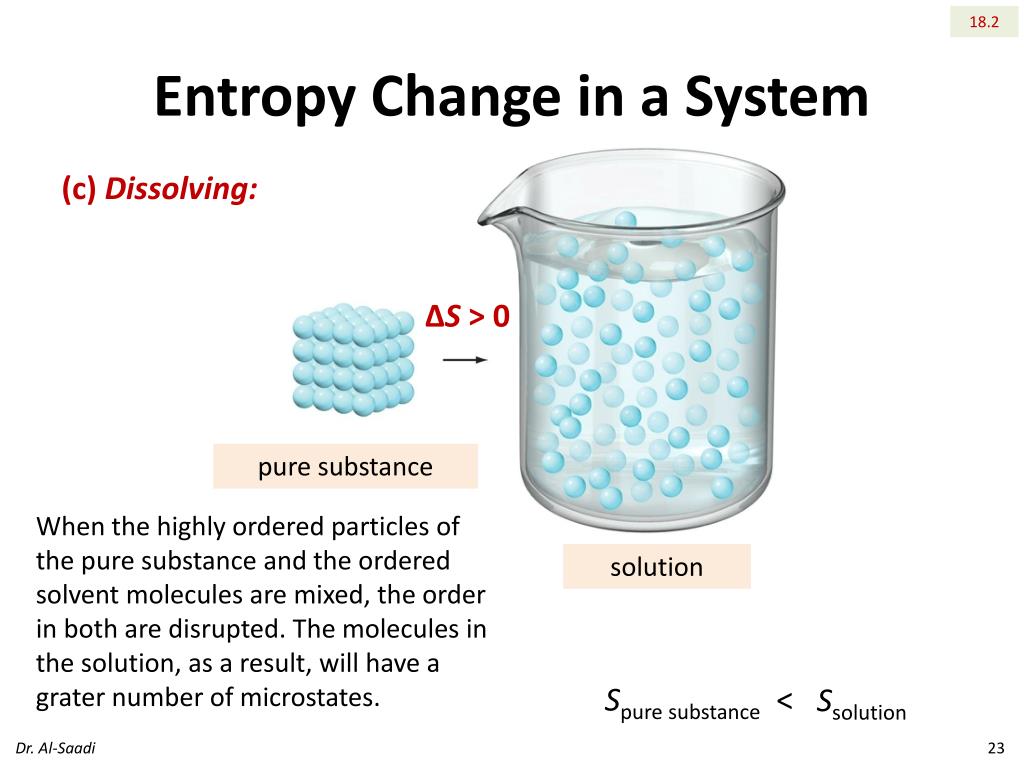
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
